OKR template to successfully patent a new medical device
Your OKR template
The objectives of this OKR include drafting, finalizing, and submitting the patent application within an 8-week timeline. Achieving these objectives involves careful planning, drafting of claims, creating drawings for the application, and finally, submitting the application to USPTO.
This OKR stresses the importance of ensuring that the submitted patent application is acknowledged as received by the USPTO, to be done within 12 weeks of the process. This can be confirmed by securing a confirmation of receipt from the USPTO, ensuring that the task is fulfilled.
The OKR also highlights the need to finalize the design of the device and conduct proof-of-concept tests. An initiative for these tests includes setting up a comprehensive schedule, executing the tests, compiling the results, and making any necessary final adjustments to the device design.
ObjectiveSuccessfully patent a new medical device
KRComplete comprehensive patent application, including claims and drawings, by Week 8
Draft claims and outline for patent application by Week 3
Finalise and submit patent application by Week 8
Create required drawings for application by Week 5
KRSubmit patent application to USPTO and receive confirmation of receipt by Week 12
Prepare and finalize patent application details
Submit final application to USPTO
Secure confirmation of receipt from USPTO
KRFinalize device design and conduct proof-of-concept tests by Week 4
Execute concept tests and compile results
Complete the final adjustments to device design
Set up a comprehensive schedule for concept tests
How to edit and track OKRs with Tability
You'll probably want to edit the examples in this post, and Tability is the perfect tool for it.
Tability is an AI-powered platform that helps teams set better goals, monitor execution, and get help to achieve their objectives faster.
With Tability you can:
- Use AI to draft a complete set of OKRs in seconds
- Connect your OKRs and team goals to your project
- Automate reporting with integrations and built-in dashboard
Instead of having to copy the content of the OKR examples in a doc or spreadsheet, you can use Tability’s magic importer to start using any of the examples in this page.
The import process can be done in seconds, allowing you to edit OKRs directly in a platform that knows how to manage and track goals.
Step 1. Sign up for a free Tability account
Go tohttps://tability.app/signup and create your account (it's free!)
Step 2. Create a plan
Follow the steps after your onboarding to create your first plan, you should get to a page that looks like the picture below.
Step 3. Use the magic importer
Click on Use magic import to open up the Magic Import modal.
Now, go back to the OKR examples, and click on Copy on the example that you’d like to use.

Paste the content in the text import section. Don’t worry about the formatting, Tability’s AI will be able to parse it!

Now, just click on Import from text and let the magic happen.
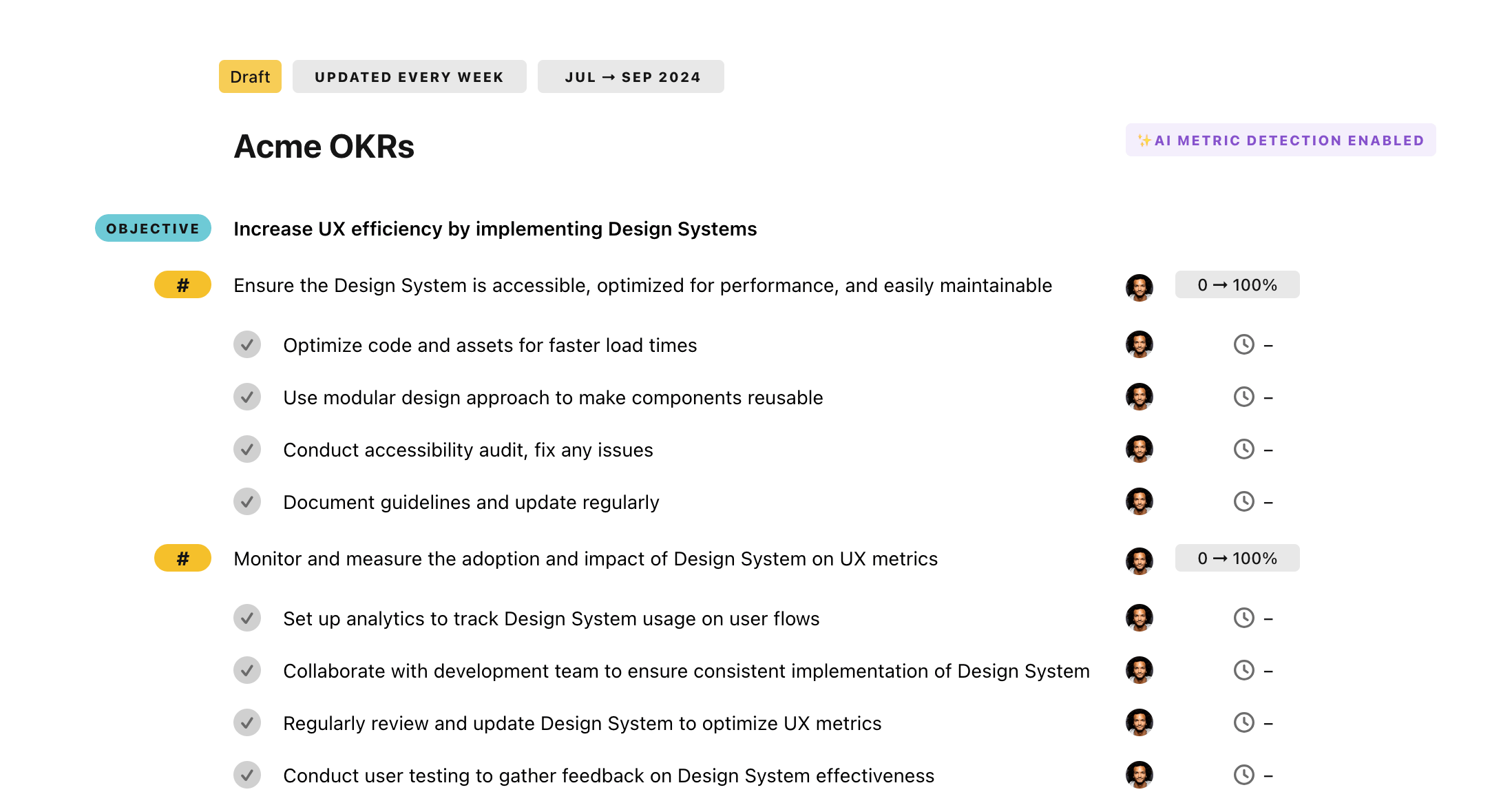
Once your example is in the plan editor, you will be able to:
- Edit the objectives, key results, and tasks
- Click on the target 0 → 100% to set better target
- Use the tips and the AI to refine your goals
Step 4. Publish your plan
Once you’re done editing, you can publish your plan to switch to the goal-tracking mode.
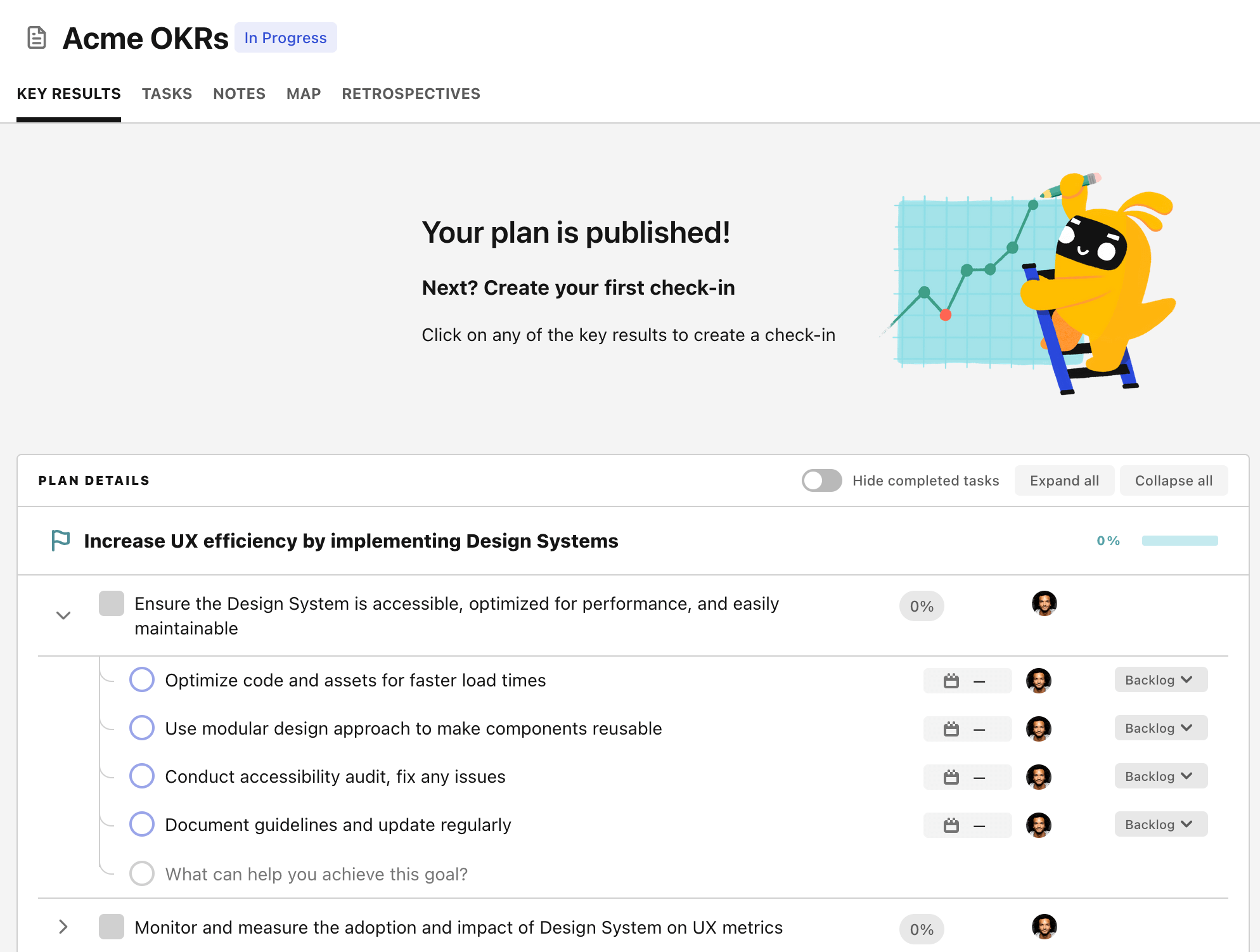
From there you will have access to all the features that will help you and your team save hours with OKR reporting.
- 10+ built-in dashboards to visualise progress on your goals
- Weekly reminders, data connectors, and smart notifications
- 9 views to map OKRs to strategic projects
- Strategy map to align teams at scale